A rule-breaking, colorful silicone that could conduct electricity. Previously only thought to be insulating, a shift in the angle between silicon and oxygen atoms creates a highway for an electrical charge. On a molecular level, silicones are made up of a backbone of alternating silicon and oxygen atoms (Si—O—Si) with organic (carbon-based) groups attached to the silicon. Various 3D formations of polymer chains arise as they connect to one another, known as cross-linking, which alter the material’s physical properties like strength or solubility.
This could lead to super-elastic silicone PV cells(/panels) and super-elastic thus super-durable, thick and highly energy dense lithium-ion battery anodes.
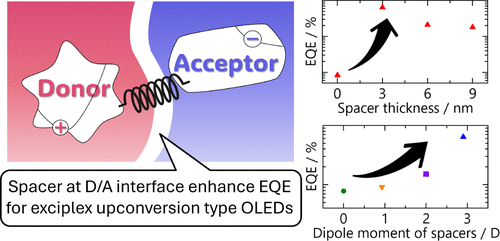
While studying different cross-linking structures in silicone, the research team stumbled upon the potential for electrical conductivity in a copolymer, which is a polymer chain containing two different types of repeating units—cage-structured and then linear silicones in this case.
The possibility for conductivity arises from the way electrons can move across Si—O—Si bonds with overlapping orbitals. Semiconductors have two main states: the ground state, which doesn’t conduct electricity, and a conducting state, which does. The conducting state, also known as an excited state, occurs when some electrons jump up to the next electron orbital, which is connected across the material like a metal.
Typically, Si—O—Si bond angles don’t allow for that connection. At 110°, they are a long way from a 180° straight line. But in the silicone copolymer the team discovered, these bonds started out at 140° in the ground state—and they stretch to 150° in the excited state. This was enough to create a highway for electrical charge to flow.
σ–σ* conjugation Across Si─O─Si Bonds. Macromol.
“This allows an unexpected interaction between electrons across multiple bonds including Si—O—Si bonds in these copolymers,” Laine said. “The longer the chain length, the easier it is for electrons to travel longer distances, reducing the energy needed to absorb light and then emit it at lower energies.”
The semiconducting properties of the silicone copolymers also enable its spectrum of colors. Electrons jump between the ground and excited states by absorbing and emitting photons, or particles of light. The light emission depends on the length of the copolymer chain, which Laine’s team can control. Longer chain lengths mean smaller jumps and lower energy photons, giving the silicone a red tint. Shorter chains require bigger jumps from the electrons, so they emit higher energy light toward the blue end of the spectrum.
To demonstrate the connection between chain length and light absorption and emission, the researchers separated copolymers with different chain lengths and arranged them in test tubes from long to short. Shining a UV light on the tubes creates a full rainbow as each absorbs and emits the light at different energies.
The colorful array based on copolymer chain length is particularly unique because up to this point, silicones have only been known to be transparent or white because their insulating properties make them unable to absorb much light.

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.